Cell-Penetrating Peptide Enhances Tafazzin Gene Therapy in Mouse Model of Barth Syndrome
Authors: Rahul Raghav, Junya Awata, Gregory L. Martin, Douglas Strathdee, Robert M. Blanton, Michael T. Chin
Journal: International Journal of Molecular Sciences (link to article)
Background:
Barth Syndrome is caused by a change (or mutation) in the DNA segment that produces the protein TAFAZZIN; changes to the TAFAZZIN sequence frequently cause the protein to lose its function. Normally, the job of TAFAZZIN protein is to convert a special fat or lipid called monolysocardiolipin (MLCL) into its mature form called cardiolipin (CL). In Barth Syndrome, loss of TAFAZZIN function prevents cells from making CL and instead, results in too much MLCL and too little CL. This is important because a build-up of MLCL and too little CL reduces the function of mitochondria, which are special structures within almost every cell in the body whose main job is to make energy. The heart requires a lot of energy to function properly, so a build-up of MLCL decreases heart function. The build-up of MLCL compared to CL is a diagnostic hallmark in Barth syndrome and can be used to help doctors determine if an individual has Barth syndrome.
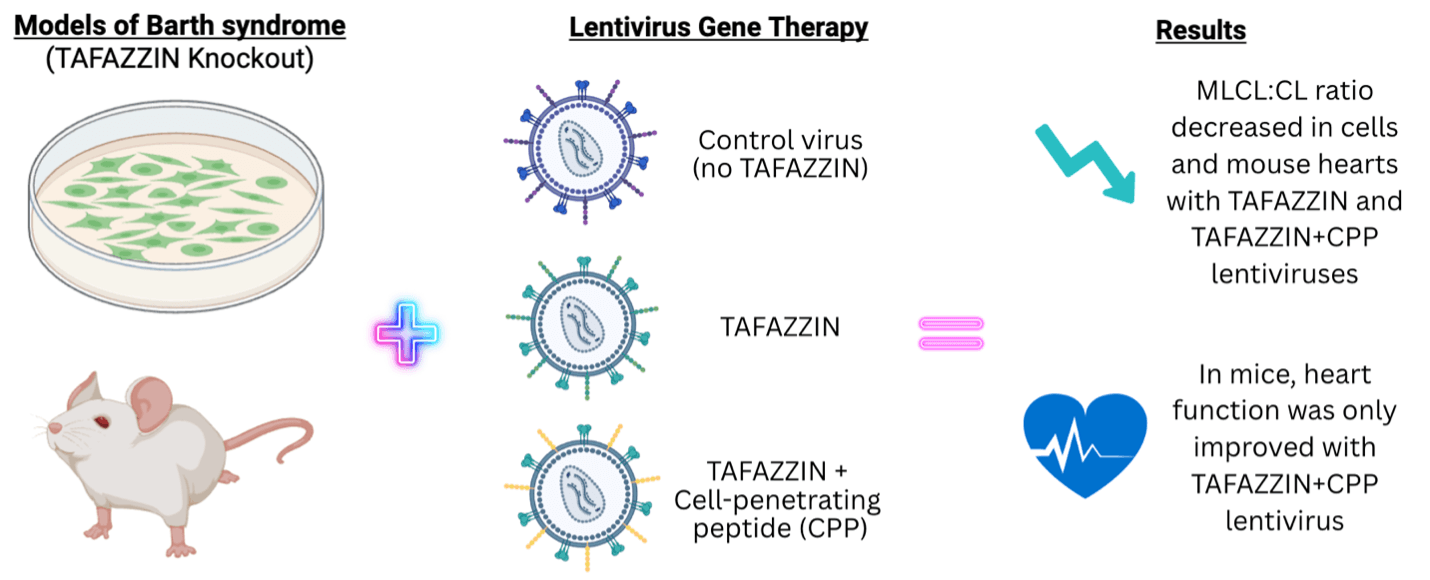
Gene addition therapy is a type of treatment that aims to deliver a functional version of TAFAZZIN DNA into cells that have non-functional TAFAZZIN. TAFAZZIN DNA can be delivered into the body using special types of viruses, such as lentiviruses or adeno-associated viruses, as a transport method. You can think of this system like a spaceship being launched, where the goal is for the spaceship (virus) to deliver its cargo (functional TAFAZZIN) to cells that need it most (heart and skeletal muscle). These viruses are engineered such that they don’t cause virus-related diseases but rather serve just as delivery vehicles. The goal of the present study by Dr. Michael Chin and colleagues was to evaluate whether the effectiveness of lentivirus gene addition therapy could be improved by adding an extra sequence called a cell-penetrating peptide (CPP). CPPs are special protein fragments that can be added onto another protein (in this case, TAFAZZIN) to enhance entry into cells.
Findings:
The researchers first tested the TAFAZZIN lentiviral therapy in two models of Barth syndrome: cells and mice. The researchers compared three different treatment groups:
(1) TAFAZZIN lentivirus
(2) TAFAZZIN+CPP lentivirus or
(3) a control lentivirus without the TAFAZZIN gene.
Cells were treated with lentivirus and examined after a week of treatment. To determine how the treatment affected the cells, the researchers measured the ratio of MLCL to CL. Treatment with TAFAZZIN or TAFAZZIN + CPP decreased the MLCL:CL ratio to normal levels and no differences were discernable when CPP was added; the control virus did not improve the MLCL:CL ratio. This indicates that addition of the CPP did not further enhance TAFAZZIN protein function in a cell model of Barth syndrome. The researchers then tested their lentiviral therapies in a mouse model of Barth syndrome. In this model, they looked at the MLCL:CL ratio as well as heart function using echocardiography (interventricular septal thickness, ejection fraction, fractional shortening, and global longitudinal strain). Interestingly, while both the TAFAZZIN and TAFAZZIN+CPP lentiviruses equally reduced the MLCL:CL ratio, only the TAFAZZIN+CPP was able to improve heart function in the Barth mouse model, suggesting that addition of a CPP could be beneficial for Barth-targeted gene therapies.
Take-Home Message:
This study provides evidence that addition of a cell-penetrating peptide to TAFAZZIN can improve the effectiveness of a gene therapy for treating Barth Syndrome. This finding is important because previous research has contributed to a hypothesis that improvement of the MLCL:CL ratio should correlate with improved heart function. Future research is required to understand how the addition of the CPP improved both the MLCL:CL ratio and heart function.



